ICMR issues revised advisory on use of hydroxychloroquine; check details
New Delhi: The Indian Council of Medical Research (ICMR) on Saturday used a revised guideline on who can use the Hydroxychloroquine.
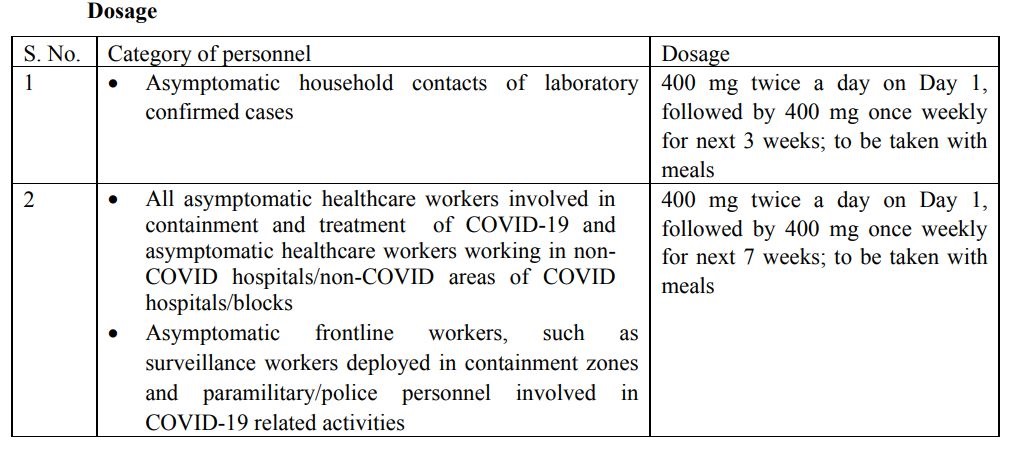
As per the new guideline, all asymptomatic healthcare workers involved in containment and treatment of COVID19 and asymptomatic healthcare workers working in non-COVID hospitals/non-COVID areas of COVID hospitals/blocks can use Hydroxychloroquine.
Likewise, the asymptomatic frontline workers, such as surveillance workers deployed in containment zones and paramilitary/police personnel involved in COVID-19 related activities also can use it
Moreover, asymptomatic household contacts of laboratory confirmed cases also have been permitted to use Hydroxychloroquine.
How much doge can be used?
Exclusion/contraindications
- The drug is contraindicated in persons with known case of: 1. Retinopathy, 2. Hypersensitivity to HCQ or 4-aminoquinoline compounds 3. G6PD deficiency 4. Pre-existing cardiomyopathy and cardiac rhythm disorders
- The drug is not recommended for prophylaxis in children under 15 years of age and in pregnancy and lactation. Rarely the drug causes cardiovascular side effects such as cardiomyopathy and rhythm (heart rate) disorders. In that situation the drug needs to be discontinued. The drug can rarely cause visual disturbance including blurring of vision which is usually self- limiting and improves on discontinuation of the drug. For the above cited reasons the drug has to be given under strict medical supervision with an informed consent
Monitoring
- An ECG (with estimation of QT interval) may be done before prescribing HCQ prophylaxis.
- An ECG should be done in case any new cardiovascular symptoms occurs (e.g., palpitations, chest pain syncope) during the course of prophylaxis.
- An ECG (with estimation of QT interval) may be done in those who are already on HCQ prophylaxis before continuing it beyond 8 weeks.
- One ECG should be done anytime during the course of prophylaxis.
Key considerations
While following above recommendations, it should be noted that:
1) The drug has to be given under strict medical supervision with an informed consent.
2) The drug has to be given only on the prescription of a registered medical practitioner.
3) Advised to consult with a physician for any adverse event or potential drug interaction before initiation of medication. The contraindications mentioned in the recommendations should strictly be followed.
4) Health care workers and other frontline workers on HCQ should be advised to use PPE. Front line workers should use PPEs in accordance with the guidelines issued by this Ministry or by their respective organization.
5) They should be advised to consult their physician (within their hospital/surveillance team/security organization) for any adverse event or potential drug interaction before initiation of medication. The prophylactic use of HCQ to be coupled with the pharmacovigilance for adverse drug reactions through self-reporting using the Pharmacovigilance Program of India (PvPI) helpline/app.
6) If anyone becomes symptomatic while on prophylaxis, he/she should immediately contact the health facility, get tested as per national guidelines and follow the standard treatment protocol. Apart from the symptoms of COVID-19 (fever, cough, breathing difficulty), if the person on chemoprophylaxis develops any other symptoms, he should immediately seek medical treatment from the prescribing medical practitioner.
7) All asymptomatic contacts of laboratory confirmed cases should remain in home quarantine as per the National guidelines, even if they are on prophylactic therapy.
8) Simultaneously, proof of concept and pharmacokinetics studies should be continued/ taken up expeditiously. Findings from these studies and other new evidence will guide any change further in the recommendation.
9) They should follow all prescribed public health measures such as frequent washing of hands, respiratory etiquettes, keeping a distance of minimum 1meter and use of Personal protective gear (wherever applicable).



 Ms Kalinga
Ms Kalinga